
MAE graduate research assistant Suhas Tamvada has made a major discovery in the field of heat transfer, a breakthrough that addresses a question debated in nucleate boiling science for decades. The research and its findings have recently appeared in the International Journal of Heat and Mass Transfer.
Tamvada’s research, conducted alongside UF MAE’s Dr. Saeed Moghaddam and scientific consultant Daniel Attinger, owner of Struo LLC, addressed the two prevailing theories regarding critical heat flux (the maximum amount of heat that can be transferred from a surface to a liquid while boiling). The proposed theories on the existence of this limit had to do with hydrodynamic instability (a phenomenon happening away from the boiling surface) and evaporation momentum force theory (happening on the surface itself). These two theories predicted roughly similar critical heat flux values, but the team’s paper shows that the evaporation momentum limit is 3 to 4 times higher than previously thought. Tamvada’s paper describes how, though these are two contrasting phenomena, they both are present throughout the entire boiling process. However, his research showed that, provided with the right experimental conditions, it is possible to overcome the limit imposed by the hydrodynamic instability and establish a flow condition wherein the evaporation momentum limit can be observed as the ultimate limit.
“That has a lot of implications,” he said. “Now people can start rethinking the entire surface design for different applications, because now the physics is no longer ambiguous.”
Critical heat flux is a crucial factor in many real-world applications, such as cooling of nuclear reactors, data centers, and power electronics. For example, the capacity of a nuclear power plant is determined by the maximum heat flux that can be removed from the nuclear fuel rods. Increasing the critical heat flux limit would allow for more energy to be extracted from the fuel rods.
Researchers have been trying to decipher the underlying physics of the critical heat flux phenomenon since the limit was discovered in 1934 by Shiro Nukiyama.
“Figuring out the physics behind the critical heat flux phenomenon has been a dream of mine for 20 years,” Moghaddam said. “This is truly a gratifying moment for me.”
Tamvada said that the genesis of the project was about starting with the source of the problem, which, according to him, was that “nobody was able to design a set of experiments which could show, conclusively, that one limit is fully surpassed such that the other limit can be observed.” In 2017, Moghaddam’s group discovered that separating liquid and vapor flow above a hot surface with the help of a membrane results in a unique heat flux value of 400 to 500 watts per centimeter squared on five completely different surfaces when normalized by the total surface area of the heater.
Moghaddam hypothesized that this unique critical heat flux value observed among different surfaces is a surface phenomena consistent with the evaporation momentum theory (the work was featured in Nature Scientific reports). Subsequently, Tamvada and Moghaddam deliberated methods to design an experiment that would unambiguously settle the debate on the mechanism of critical heat flux.
“Having, I would say, courage on our part to question existing belief systems was the key reason behind our success,” Tamvada said.
He attributes this success partly to the group’s commitment to try methods that hadn’t been considered before.
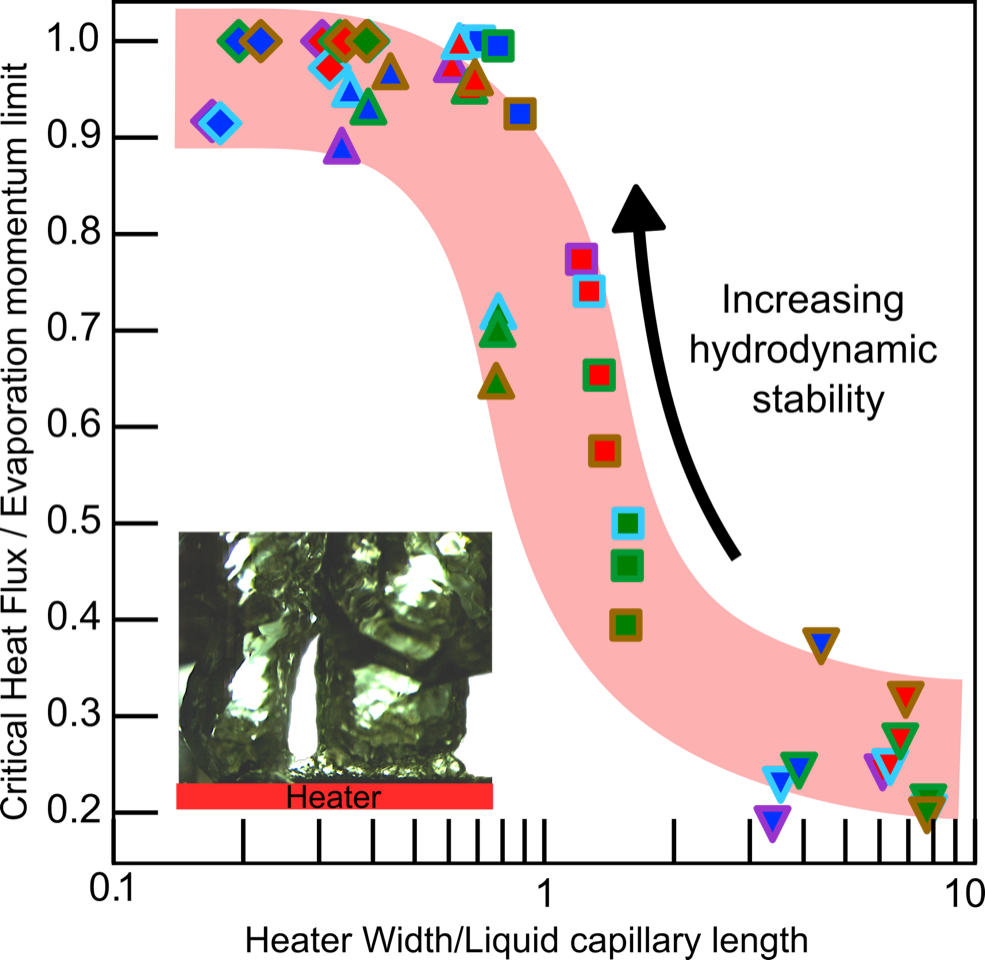
“Although researchers had previously moved in the right direction in 1973 (Lienhard and Dhir) by reducing heater size, they could not conclude that the CHF they observed was surface limited since the surface phenomena was only formalized in the late 90s by a group in the UK, when they proposed the evaporation momentum theory (Steinchen and Sefiane)” he said. “After the hydrodynamic limit was proposed, people tried different combinations of surfaces and liquids, and they did see some improvement in the critical heat flux, but nobody ever pushed the limit and went below something that we call the capillary length of a liquid. We designed experiments keeping in mind that in order to observe a surface limited phenomenon, we had to overcome the limit imposed by hydrodynamic instabilities. So, we fabricated heater surfaces around the capillary lengths of the liquids we wanted to test.”
The capillary length is a characteristic length of a liquid signifying the relative importance of the buoyancy and capillary forces acting on the liquid. One of the principal findings of his research was that the capillary length of a liquid plays a major role in determining whether it’s limited by hydrodynamics or whether it’s limited by evaporation momentum.
Moghaddam’s praise for Tamvada was effusive and full of gratitude.
“Students are down in the trenches and ultimately make the magic happen,” he said. “I have been blessed with having a student like Suhas. He is capable, excited, and dedicated. He gives me energy, and I always look forward to my next intellectual conversation with him.”
This research is supported by Award No. CBET 1934354 from the United States National Science Foundation (NSF).
Story by: Ben Crosbie
Marketing & Communications Student Assistant
UF Mechanical & Aerospace Engineering
March 23, 2023
